ProLon® Just Five Days, You Won’t Be disappointed! | El Paso, TX.
Recent science has unleashed the power of fasting to optimize body aging and dismantle chronic diseases, e.g., obesity, diabetes, cancer, autoimmune disorders, Alzheimer’s and cardiovascular disease. At PUSH-as-Rx ®™, Dr. Alex Jimenez’s mission is to educate the public about the health benefits of fasting. The idea is to introduce it to them in a safe, effective way through the Fasting Mimicking Diet (FMD).
Pre-clinical and clinical studies have proven that periodic fasting, done for several consecutive days, is a very powerful intervention that our bodies learned to naturally cope with by protecting and rejuvenating itself. These two factors are both anti-aging measures that offer additional health benefits. The 5-Day ProLon Fasting Mimicking Diet has been clinically tested and found to promote beneficial effects in a wide variety of conditions ranging from excess weight and fasting blood glucose, to growth factors associated with DNA damage and aging.
Min Wei; Sebastian Brandhorst et al.
Fasting?Mimicking Diet and Risk Factors for Aging, Diabetes, Cancer and Cardiovascular Disease.
Table of Contents
5-Day Program
The ProLon meal plan is followed 5 days per month. Once an individual has finished the five-day plan, they go back to a normal healthy diet the last twenty-five days. Fasting with the Prolon® plan follows a low carbohydrate/protein meal and contains the good kind of fatty acids. The FMD® recipe keeps your body on a fasting-type mode, that triggers protection measures that the body has developed. This causes the body to optimize its performance, rejuvenate cells, and thrive.
Unboxing the ProLon Fasting Mimicking Diet

Age-related Disease: A Revolution Is Coming
I just finished reading a groundbreaking book called The Longevity Diet: Discover the New Science Behind Stem Cell Activation and Regeneration to Slow Aging, Fight Disease, and Optimize Weight, which was written by Valter Longo, PhD,1 who is director of the Longevity Institute at the University of Southern California and a principal scientist in the development and study of the fasting mimicking diet (FMD). I have followed Dr. Longo’s career for many years with great admiration. He has been published in top-tier journals. In his book, Dr. Longo writes about some exciting findings regarding the FMD research group consisting of 16-month-old mice, which are described as being the equivalent of a 45-year-old human: “A stem cell-dependent process rejuvenated the immune system. Regeneration also occurred in the liver, muscle, and brain. Levels of several types of stem cells increased.”1 He went on to explain: “The fasting itself destroys many damaged cells and damaged components inside the cells but it also activates stem cells.”1
Jeffrey S. Bland, PhD, FACN, FACB, Associate Editor
Get Your Free Copy Today!
Fasting-Mimicking Diet Promotes b-Cell Regeneration to Reverse Diabetes
- Fasting mimicking diet induces prenatal-development gene expression in adult pancreas
- FMD promotes Ngn3 expression to generate insulin-producing b cells
- Cycles of FMD reverse b-cell failure and rescue mice fromT1D and T2Dd
- Inhibition of PKA or mTOR promotes Ngn3-driven b-cell regeneration in human T1D islets
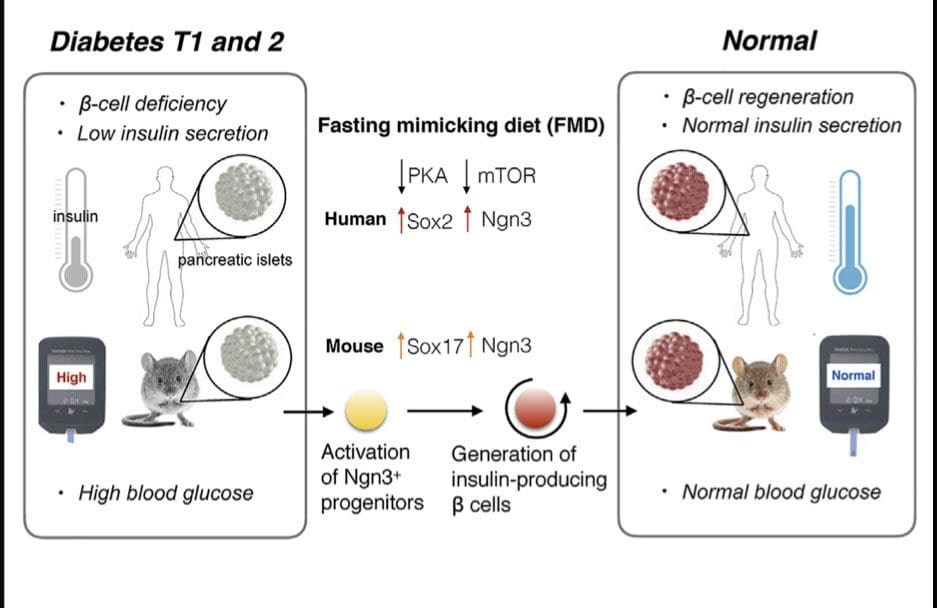
Stem-cell-based therapies can potentially reverse organ dysfunction and diseases, but the removal of impaired tissue and activation of a program leading to organ regeneration pose major challenges. In mice, 4-day fasting mimicking diet (FMD) induces a stepwise expression of Sox17 and Pdx-1, followed by Ngn3-driven generation of insulin-producing b cells, resembling that observed during pancreatic development. FMD cycles restore insulin secretion and glucose homeostasis in both type 2 and type 1diabetes mouse models.
In human type 1 diabetes pancreatic islets, fasting conditions reduce PKAand mTOR activity and induce Sox2 and Ngn3expression and insulin production. The effects of the FMD are reversed by IGF-1 treatment and recapitulated by PKA and mTOR inhibition. These results indicate that an FMD promotes the reprogramming of pancreatic cells to restore insulin generation in islets from T1D patients and reverse both T1D andT2D phenotypes in mouse models.
Chia-Wei Cheng,1,6,7Valentina Villani,2,7Roberta Buono,1,5,7Min Wei,1Sanjeev Kumar,4Omer H. Yilmaz,6Pinchas Cohen,1Julie B. Sneddon,3Laura Perin,2and Valter D. Longo1,4,5,8,*
Post Disclaimer *
Professional Scope of Practice *
The information herein on "ProLon® Just Five Days, You Won't Be disappointed! | El Paso, TX." is not intended to replace a one-on-one relationship with a qualified health care professional or licensed physician and is not medical advice. We encourage you to make healthcare decisions based on your research and partnership with a qualified healthcare professional.
Blog Information & Scope Discussions
Our information scope is limited to Chiropractic, musculoskeletal, physical medicines, wellness, contributing etiological viscerosomatic disturbances within clinical presentations, associated somatovisceral reflex clinical dynamics, subluxation complexes, sensitive health issues, and/or functional medicine articles, topics, and discussions.
We provide and present clinical collaboration with specialists from various disciplines. Each specialist is governed by their professional scope of practice and their jurisdiction of licensure. We use functional health & wellness protocols to treat and support care for the injuries or disorders of the musculoskeletal system.
Our videos, posts, topics, subjects, and insights cover clinical matters, issues, and topics that relate to and directly or indirectly support our clinical scope of practice.*
Our office has reasonably attempted to provide supportive citations and has identified the relevant research study or studies supporting our posts. We provide copies of supporting research studies available to regulatory boards and the public upon request.
We understand that we cover matters that require an additional explanation of how it may assist in a particular care plan or treatment protocol; therefore, to further discuss the subject matter above, please feel free to ask Dr. Alex Jimenez, DC, or contact us at 915-850-0900.
We are here to help you and your family.
Blessings
Dr. Alex Jimenez DC, MSACP, RN*, CCST, IFMCP*, CIFM*, ATN*
email: coach@elpasofunctionalmedicine.com
Licensed as a Doctor of Chiropractic (DC) in Texas & New Mexico*
Texas DC License # TX5807, New Mexico DC License # NM-DC2182
Licensed as a Registered Nurse (RN*) in Florida
Florida License RN License # RN9617241 (Control No. 3558029)
Compact Status: Multi-State License: Authorized to Practice in 40 States*
Presently Matriculated: ICHS: MSN* FNP (Family Nurse Practitioner Program)
Dr. Alex Jimenez DC, MSACP, RN* CIFM*, IFMCP*, ATN*, CCST
My Digital Business Card





